Iron and Steel Works
Iron and Steel Works and Stockholders
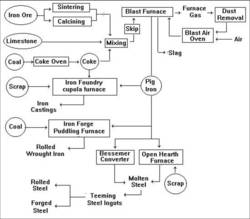
Recovering metals from ore is called smelting, this is usually done by heating the ore to the melting point of the metal in a blast furnace. This drives off some of the impurities and others burn in the furnace to form gasses, meanwhile the required metal melts and this can be separated from other molten materials (slag) by settling them out. As shipping the low value ore is relatively more expensive than moving the high value pure metal the smelting areas were in the main located close by the mines. There were exceptions however, the South Wales tin plate and copper smelting firms had plentiful supplies of good quality coal close to hand and used ore carried cheaply by sea from Cornwall. Also in some cases the local industry was sufficiently strong to justify the movement of ore or partially refined metal when the local supplies ran out. A good example of the latter is the carriage of pig iron from Cleveland to the iron works in Staffordshire and Shropshire in the later nineteenth century to allow these works to continue working when their own deposits had been worked out. General History of the Iron and Steel making process Iron is probably the most important metal known to man, without it life as we know it today could not exist. Although other metals are used in considerable quantity, notably aluminium, copper, tin, lead and zinc, iron is still the most important. Iron possesses magnetic properties which allowed the development of electrical power. Both iron and steel are the same material but in the latter more of the impurities (mainly carbon) are removed. The British Industrial Revolution, the first in the world, was based on iron, the rest of the world industrialised later and based their machines on steel. Many of the processes involved in smelting iron and making steel required chambers lined with heat resistant bricks. Although heat resistant these bricks are not heat proof and they had to be replaced at intervals (a dirty job by all accounts), hence wagon loads of bricks would be a feature of both iron and steel works. The fire bricks are XXX in colour. XXX More on fire bricks XXX Fig ___ Iron & Steel Making Flow Diagram
Iron was discovered thousands of years ago but early extraction methods relied on charcoal made from wood which was time consuming, expensive and only available in small quantities. Up to 1709 charcoal was used as a fuel for the blast furnaces which separate the iron from the oxygen and other impurities.Abraham Darby then developed the use of coke in place of charcoal, he did not protect the process with patents and this allowed the rapid development of the iron industry. In Darby's time British iron production was running at 17,000 tons a year, by the 1950's it was up to over fifteen million tons a year. The 'pig iron' produced in the blast furnace has a high carbon content and cannot practically be used for very much. if however it is re-melted more impurities can be removed. This re-melting is usually done in a small scale blast furnace called a 'cupola furnace' (invented in the 1790's by a British ironmaster called John Wilkinson). Scrap metal can be recycled in this furnace along with the pig iron. The molten iron which comes out of a cupola furnace flows well and can be used to make solid castings, it is called 'cast iron'. Cast iron contains a lot of carbon and tends to be brittle, it serves well under compression but is poor when placed under tension. Places which take in pig iron to make cast iron are called 'foundries', you would normally have a foundry associated with an iron works but there were also small establishments catering to local industry (most towns had at least two small foundries) and larger engineering works often had their own foundry on-site.
Pig iron can be used to make cast iron goods in a foundry (see also Lineside Industries - Scrap metal yards, Foundries and Forges) but this material, although resistant to rusting and strong under compression, is brittle and cannot be re-heated and shaped. Cast iron is still widely used today (motor car engine blocks are made of cast iron).
If you remove nearly all the carbon from pig iron or cast iron you get 'wrought iron' (sometimes called 'malleable iron' in older texts) which contains less than 0.1% carbon and about 3% 'slag'. Wrought iron is much stronger under tension than cast iron and the slag forms a coating or skin on the surface which imparts certain qualities to the metal, notably corrosion resistance. Wrought iron can be hammered into shape when red hot so places where wrought iron was made are usually called 'forges' (a forge is a hammer and an anvil). Originally wrought iron was made by beating bars of red hot cast iron and beating it to de-form it and allow the air to get at more of the carbon (which then burns away). This was done in a works known as a 'finery'.
In 1784 an iron works owner by the name of Henry Cort obtained a patent for producing wrought iron in bulk by stirring molten iron in a reverbatory furnace (a process known as 'puddling', for a description of a reverbatory furnace see Lineside Industries - Prototype industrial ancillary structures). What happens is that the air would oxidise and remove some of the remaining carbon (becoming CO2 gas) producing low carbon wrought iron. Mr Cort also developed and patented a hot rolling process for iron heated until it was the consistency of a thick paste and then passed between powered rollers in a rolling mill which produced better quality iron much faster. The combination of these two developments allowed his works to produce about fifteen times as much iron in a given time and with a given amount of fuel. Cort's business partner was then prosecuted for fraud and (as he was a full partner) Cort was also held responsible and his patents (both for the puddling furnace and his wrought iron rolling mills) were confiscated and thrown open to all manufacturers, the result was a massive increase in wrought iron production. The combination of cast and wrought iron along with Mr Cort's rolling mill allowed the industrial revolution to mechanise production but by the 1870's iron production had peaked and the industry went into slow decline as steel began to become the more significant material.
Cort's puddling process was further improved in 1816 when Joseph Hall of Tipton developed an oxygen rich lining for the puddling furnace (this was called 'wet puddling' to differentiate it from Cort's system which in turn became known as 'dry puddling'). A small quantity of haematite ore is added to the pigs of iron in the cupola at the foundry. The oxygen in the haematite (Fe3O2) combines with most of the carbon in the pig iron to form carbon dioxide and the resulting iron has less than 0.1% carbon in it.
Wrought iron can be heated and rolled into long strips or sheets. The first sheet rolling mill was built in
Wrought iron can also be formed to shape by beating it and belting it with a hammer or pressing it over a former. This heating and forming with hammers and presses is called forging. A forge consists of a solid base and a hammer, a blacksmith's forge had the anvil and the blacksmith used a hand held hammer. The early 'industrial' forges used water-power to drive the hammer, steam powered hammers were invented in
Steel is iron with all the carbon removed and a little added back in, this produces something chemically similar to wrought iron but with a different crystalline structure. Up to the middle of the nineteenth century it was made in small pots (called 'crucible steel') and prices reflected the small scale and labour intensive manufacture. By the 1850's
The next big development was the Siemens-Martin or open hearth process of 1866, using the reverbatory regenerative furnace invented by Frederick Siemens (1826-1904) in 1856. The regenerative furnace uses the waste heat generated to pre-heat incoming air instead of simply blowing it out of the top as in the
In 1863 Henry Clifton Sorby (1826-1908) working in
The Basic Oxygen Process for making steel was developed in Austria in about 1950, and this system has turn largely replaced the open hearth system. The Basic Oxygen Process uses pure oxygen in large quantities (called 'tonnage oxygen') which is blown into the liquid metal to combine with the carbon and remove it (lime can also be injected to reduce the phosphorous content). The oxygen used for this system is usually made on site but when the on-site equipment is being serviced oxygen is delivered in British Oxygen Company liveried cryogenic (low temperature) railway tank wagons.
There is one other option for making steel, the electric furnace process, which offers the possibility of turning the heat up or down and thus controlling directly the creation of the steel. It costs a lot to make steel in this way so the technique is only used for making high grade alloy steels such as stainless steels and steel used for cutting tools.
By 1968 technology had improved to the point where molten steel could be poured into the top of a water cooled mould and drawn out as a continuous bar at the bottom. This technology was soon adopted in most of the worlds major steel works.
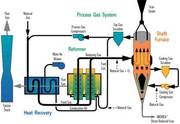
STEEL INDUSTRY IN IRAN
Notifying Visitors of Site Enhancements
Need some extra help building your site? Here are some topics that may be helpful.